Intracellular rheology for cell elasticity and viscosity measurements
Intracellular active rheology
Probe the strain response of intracellular medium as a function of frequency with our automatized TimSom Active Rheology routine and obtain the storage (G′, elastic contribution) and loss (G″, viscous contribution) moduli of cell cytoplasm.
Quantify cell stiffness, viscosity and elasticity
Our SENSOCELL optical tweezers allow measuring the rheological properties of cells inside tissues, embryos and living organisms such as Zebra fish or C. elegans systems.
Use intracellular endogenous organelles as probes
Perform active rheology assays of cells trapping either internalized microspheres or endogenous organelles such as vesicles or lipid droplets as probes.
Frequency spectrum of G modulus over 5 orders of magnitude
The Sensocell optical tweezers workflow allows applying oscillations to the trapped probes with frequencies ranging from 0.01 Hz up to 6 kHz and automatically computes the complex shear modulus G of the sample at the selected frequencies.
Selected publications:
- Hurst, S., Vos, B.E., Brandt, M. et al. Intracellular softening and increased viscoelastic fluidity during division. Nat. Phys. 17, 1270–1276 (2021).
- Kechagia, Z., Sáez, P., Gómez-González, M. et al. The laminin–keratin link shields the nucleus from mechanical deformation and signalling. Nat. Mater. 22, 1409–1420 (2023)
- Frederic Català-Castro, Santiago Ortiz-Vásquez, Carmen Martínez-Fernández et al. Active microrheology with a single, time-shared laser trap. Biorxive 2023
Application Note:
Micro-rheology of soft biological samples using SENSOCELL optical tweezers.
Related applications:
Cytoplasm and nucleus rheology inside a Zebra fish progenitor cell
In this example, the SENSOCELL optical tweezers were used to measure the viscoelastic properties of Zebra fish stem cells ex vivo. The researchers used the TimSOM active rheology routine to obtain the complex shear modulus G at different locations of the cell: at the cell’s cytoplasm, the nuclear interface and inside the cell nucleus.
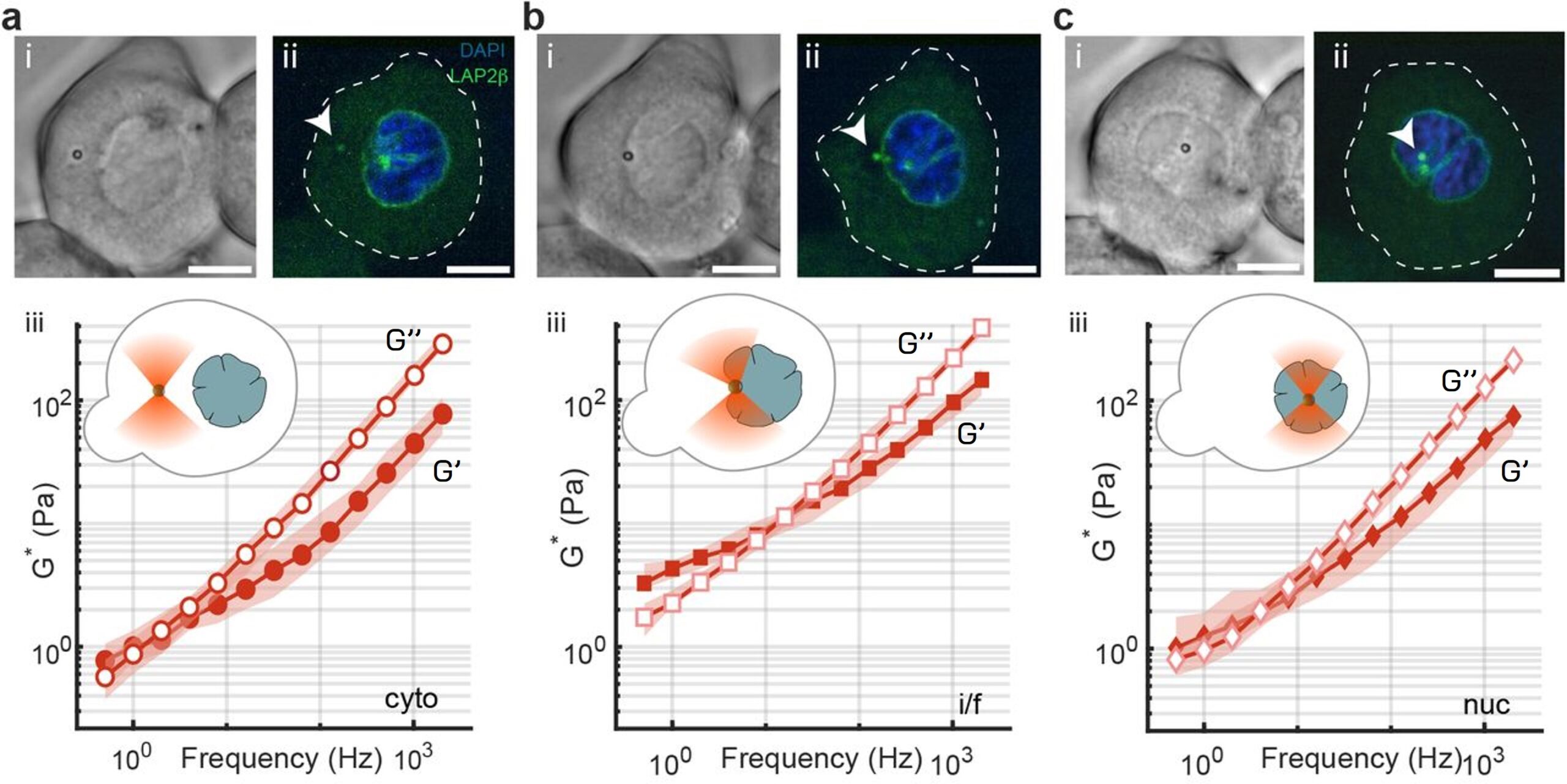
Fig. 1 Brightfield (i) and confocal (ii) images of an isolated Zebrafish progenitor cell with an internalized microsphere used as optical trapping probe for the intracellular rheology measurements performed with Sensocell at the cell’s cytoplasm (a) the nuclear interface (b) and inside the nucleus (c). Below, the frequency spectrum of the complex shear modulus (iii) of the corresponding probed medium is plotted indicating the storage and loss moduli contributions. Scale bar is 10 μm. Courtesy of M. Krieg’s lab (ICFO).
In vivo cytoplasm & nucleus rheology inside a living c. elegans organism
Quantifcation of the complex shear modulus of Caenorhabditis elegans changes during age using TimSOM Active Rheology with Sensocell optical tweezers. Using mutants known to display premature ageing phenotypes in C. elegans, M. Krieg’s group revealed how mutations causing nuclear envelopathies soften the cytosol of intestinal cells during organismal age. The data shows the frequency dependent shear modulus for two different ages of wildtype and age-matched lem-2 mutants and how the rheological properties of the cellular cytoplasm evolve during the first eight days of age. For these measurements, endogenous lipid droplets were used as mechanical stress probes. The viscosity of the cytoplasm was extracted from the fit of the fractional Kelvin-Voigt model to the rheological spectrum data. See more details in Biorxive 2023. Courtesy of M. Krieg’s lab (ICFO).
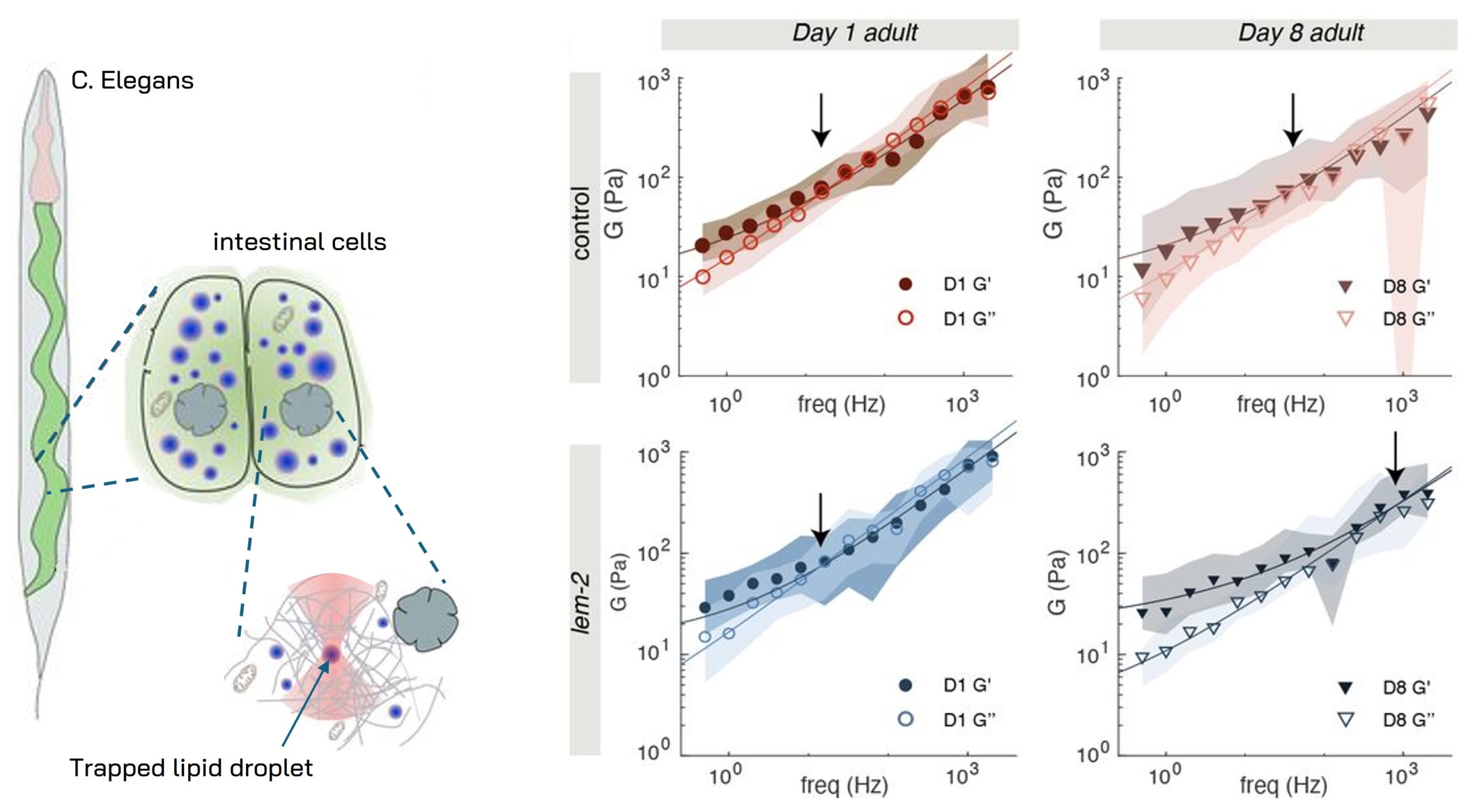
Fig. 1 Data shows the frequency dependence of the storage modulus G’ (closed symbols) and loss modulus G” (open symbols) measured inside intestinal cells of living c.elegans organisms. Left figures correspond to 1 day age and right figures to 8 days age for wildtype (top) and age-matched lem-2 mutants (bottom).
Intracellular active rheology inside a yeast cell
Full video of a TimSom active micro-rheology experiment carried out inside a living yeast cell with SENSOCELL optical tweezers. The cell’s vacuole was optically trapped and used as a probe to obtain the energy storage G’ and energy loss G” moduli of the cellular medium in the frequency range of 0.5 Hz – 1kHz. The integrated data analysis software allows fitting the data with several biophysical models like Maxwell or Kelvin-Voigt.
See other video examples:
Key Concepts
- Rheology: a field of material science that studies the flow behavior of viscoelastic fluids. In cell biophysics, it helps explain how the plasma membrane responds to tension and how stress is distributed throughout the cytoplasm during the cell cycle.
- Shear Modulus (G): describes a material’s response to shear stress. It is related to the Young’s modulus by the equation G=E/2(1+v) where v is the Poisson ratio
- Strain (ϵ): a dimensionless parameter representing the normalized deformation that an object undergoes when subjected to stress.
- Stress (σ): the amount of force per unit area exerted on an object, measured in units of pressure (Pa).
- Viscoelasticity: the property of materials to exhibit both viscous and elastic characteristics simultaneously, meaning they partially withstand stress.
Advantages
- Precision: SENSOCELL’s high-resolution measurements allow for detailed characterization of the complex shear modulus of cellular medium and its viscoelastic properties.
- Non-destructive: these experiments can be minimally invasive, preserving cell viability and function.
- Automatized routine: the TimSom active rheology routine integrated in the SENSOCELL system offers an automatized workflow for rheological assays including fitting analysis of the computed G modulus data.
- Wide spectral range: active rheology tests deliver dynamic information spanning over five decades of frequencies.
Conclusions
The TimSom active rheology routine integrated in the Sensocell optical tweezers is a unique tool for measuring the rheological properties of the intracellular medium, delivering key information of its viscoelastic properties over a wide spectral range.
Download Full Active Micro-Rheology Application Note:
Micro-rheology of soft biological samples using optical tweezers
Would you like a DEMO?
Download SENSOCELL brochure
